
Biophysical Assays

Mass Spectrometry

Human Biobank
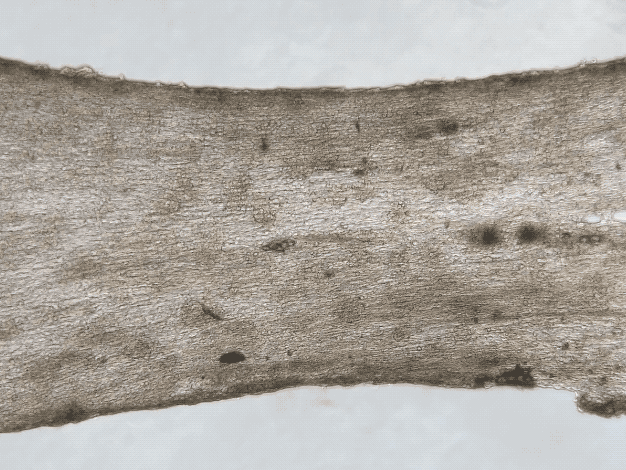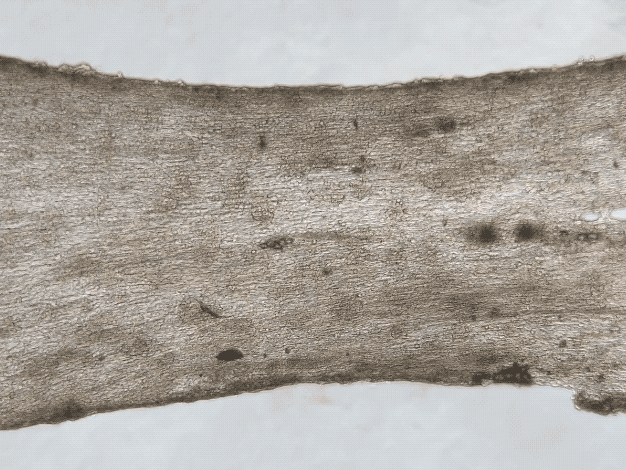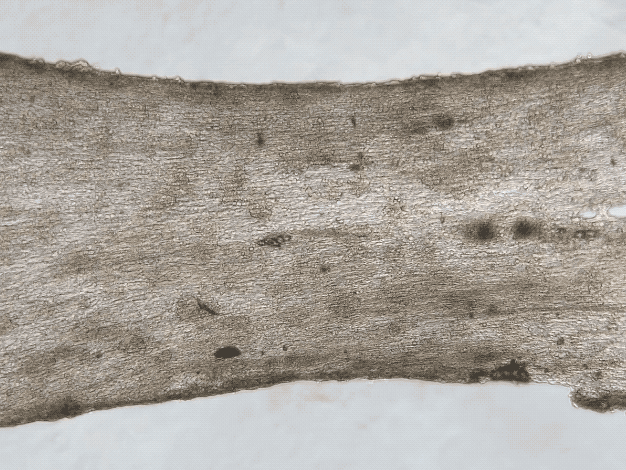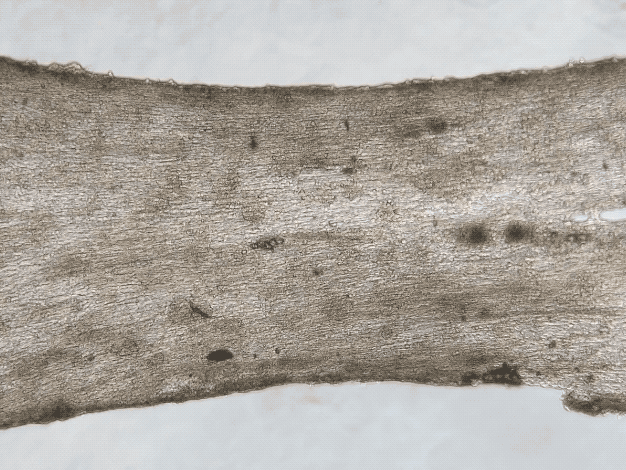
iPSC and Engineered Heart Tissues